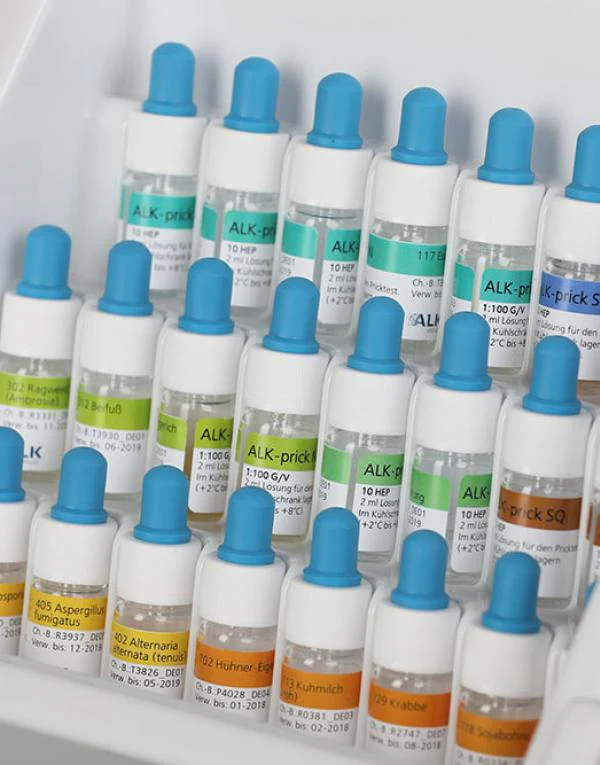
ALK bietet Ihnen repräsentative Allergen-Testlösungen für inhalative Allergene, Insektengift- und Nahrungsmittel-Allergene an.
Unser Angebot für Sie
Gebrauchs- und Fachinformationen
Die Gebrauchs- und Fachinformationen stehen Ihnen hier zum Download zur Verfügung.

Produkte
Zu den Indikationen für ALK-Präparate gehören allergischer Schnupfen, Bindehautentzündung, allergisches Asthma bronchiale sowie Reaktionen auf Bienen- und Wespenstiche. Diagnostika und ein Adrenalin-Pen runden unser Portfolio ab. Weitere Informationen und Downloads finden Sie im Login-Bereich.
Studien
ALK investiert rund 20 % seines Ertrags in Forschung, um die Allergiebehandlung stetig zu verbessern, und unterstützt Studien zur spezifischen Immuntherapie. Im Login-Bereich finden Sie aktuelle Studienergebnisse und ein umfangreiches Literaturverzeichnis.

Entdecken Sie noch mehr
Wir wollen Sie mit unseren Informationen stets auf dem aktuellen Stand halten und Sie in Ihrer täglichen Arbeit unterstützen.
Service
Im Service-Bereich finden Sie viele praktische Informationen für Ihre Praxis und Ihre Patienten.
Behandlungen
Mit einer Allergen-Immuntherapie (AIT) wird die Ursache einer allergischen Erkrankung behandelt.